Fundamentals of Fluid Flow in Porous Media
Chapter 5
Miscible Displacement
Fluid Properties in Miscible Displacement: Fluid Density
Knowledge of relative density of the fluid and fluid mixtures is important for miscible displacement design. Gravity override or underride and fingering are usual phenomena during displacement process that are results of density difference between displaced and displacing liquids. Detailed description of these phenomena will be explained in the subsequent sections. Crude oils tend to have specific gravities in the 0.80 to 0.95 range at 60°F. Density is a function of pressure and temperature and can be estimated at the reservoir condition. During the solvent injection two different fluids (oil and solvent) with different densities mix together. McCain[fusion_builder_container hundred_percent=”yes” overflow=”visible”][fusion_builder_row][fusion_builder_column type=”1_1″ background_position=”left top” background_color=”” border_size=”” border_color=”” border_style=”solid” spacing=”yes” background_image=”” background_repeat=”no-repeat” padding=”” margin_top=”0px” margin_bottom=”0px” class=”” id=”” animation_type=”” animation_speed=”0.3″ animation_direction=”left” hide_on_mobile=”no” center_content=”no” min_height=”none”][1] explained how an ideal solution behavior could be used to estimate the density of an oil-solvent mixture: An “ideal liquid solution” is a hypothetical mixture of liquids in which there is no special force of attraction between the components of the solution and for which no change in internal energy occurs on mixing. Under these conditions no change in the character of the liquids is caused by mixing, merely a dilution of one liquid by the other. So when liquids are mixed to give an ideal solution the properties are strictly additive. When the liquids mix as an ideal solution there is no shrinkage or expansion in the volume, so the final volume is the sum of the liquids volume. Other physical properties of the solution (such as viscosity and vapor pressure) can be calculated directly by averaging the properties of the fluids that mix together. There is no ideal liquid or gas solution but when the chemical and physical properties of two fluids that mix together are similar, the resulting solution behaves like as an ideal solution. McCain mentioned that because most of the liquid mixtures encountered by petroleum engineers are mixtures of hydrocarbons with similar characteristics, ideal-solution principle can be applied to find the densities of these mixtures. To find the density of a mixture that is assumed as an ideal solution it is enough to calculate the mass and volume of each of the components of the mixture and sum them together to find the mass and volume of the mixture. And then find the mixture density using these mass and volume values.
An estimate of liquid density can be calculated on the basis of a mole-averaged sensitivity of pure components as follows:

Where,
Xi = mole fraction of component I in the mixture,
ρi = Mole density of component i,
ρmix = Mole density of the mixture.
Mole density could be converted to mass density by multiplication by the average molecular weight of the mixture.
Gas density can be calculated with the EOS:

Where,
P = Pressure,
V = Specific volume,
T = Temperature,
R = Gas constant, 8.314 ( m3.Pa ) / ( K.mol )
Z = Compressibility factor that shows the deviation from ideal gas law.
Compressibility factor is a function of reduced temperature ( Tr ) and reduced pressure ( Pr ) that are defined as
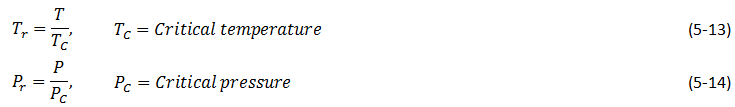
For a mixture of gases using the ideal gas mixture assumption critical pressure and temperature are defined as follow and are named pseudocritical pressure ( Ppc ) and pseudocritical temperature ( Tpc ):

Where,
n = number of components,
Xi = mole fraction of component “i”.
[fusion_separator top=”25″/]
[fusion_builder_row_inner][fusion_builder_column_inner type=”1_2″ last=”no”]
<< FLUID PROPERTIES IN MISCIBLE DISPLACEMENT
[/fusion_builder_column_inner]
[fusion_builder_column_inner type=”1_2″ last=”yes”]
[/fusion_builder_column_inner][/fusion_builder_row_inner]
[fusion_separator top=”35″/]
References
[1] “The properties of Petroleum Fluids”, McCain Jr., W.D., PennWell Publishing Co., Tulsa, OK (1973)
Questions?
If you have any questions at all, please feel free to ask PERM! We are here to help the community.[/fusion_builder_column][/fusion_builder_row][/fusion_builder_container]
